Abstract
Introduction: Myeloma is characterized by bone lesions, which are related to both an increased osteoclast activity and a defect in the differentiation of medullary mesenchymal stem cells (MSCs) into osteoblasts. Outside the medullary environment, adipocyte-derived MSCs (ASCs) could represent a source of functional osteoblasts. However, we recently found a defect in the osteoblastic differentiation of ASCs from myeloma patients (MM-ASCs). We therefore examined the effects of plasma from myeloma patients at diagnosis (MM-plasmas) and in complete remission (CR-plasmas) and from healthy donors on the osteoblastic differentiation of healthy donor-derived ASCs (HD-ASCs) and healthy donor-bone marrow derived MSCs (HD-BM-MSCs). Materials and Methods: We studied 11 MM-ASCs, 5 HD-ASCs and 3 HD-BM-MSCs. The plasmas were from myeloma patients with bone lesions at diagnosis (n=12), in complete remission (n=8) and from 5 pools of 100 healthy donors (HD-plasmas). HD-ASCs were differentiated into osteoblasts and adipocytes and HD-BM-MSCs into osteoblasts with the three types of plasmas as well as newly discovered cytokines. Results: Osteoblastogenesis in HD-ASCs was suppressed by MM-plasmas. Alizarin red coloration and alkaline phosphatase activity were strongly decreased along with a decreased RUNX2 and osteocalcin expression. However, adipocyte differentiation was unaltered. The osteoblastic differentiation deficiency was reversible once the plasma-derived factors were removed. Using cytokine array and comparing MM-plasmas with HD-plasmas, we identified seven cytokines (ANG1, ENA-78, EGF, PDGF-AA/AB/BB and TARC), besides DKK1, highly increased in MM-plasmas which was confirmed by ELISA (Figure). They separately inhibited the osteoblastic differentiation of HD-ASCs. In contrast, myeloma patients in remission had a cytokine plasma level almost normal with barely no osteoblastic differentiation inhibition. In addition, the mixture of the 7 cytokines with and without DKK1 inhibited not only the HD-ASCs but also the HD-BM-MSCs. Concomittantly, we observed that MM-plasmas enhanced adipogenesis-related gene expression. Comparison of MM-ASCs and HD-ASCs by RNA sequencing showed that two master genes characterizing adipocyte differentiation, CD36 and PPARγ, were upregulated in MM-ASCs as compared to HD-ASCs. Moreover, we demonstrated a significant increase in CD36 and PPARγ expression in HD-ASCs in the presence of MM-plasmas or the seven cytokines individually, similarly as in MM-ASCs. Finally, we tried to identify the origin of these cytokines. When myeloma patients were in remission, the cytokines levels were strongly decreased suggesting a malignant plasmocyte secretion. This was reinforced by the detection of the 7 cytokines in three different myeloma cell lines with an especially high secretion of PDGF-AA. We conclude that specific cytokines in MM-plasmas, besides the well-known DKK1, inhibit the osteoblastic differentiation of MM- and HD-ASCs with a skewing towards adipocyte differentiation. Of note, this inhibition by the cytokines were also observed on HD-BM-MSCs suggesting that this could also be the case on myeloma-BM-MSCs.
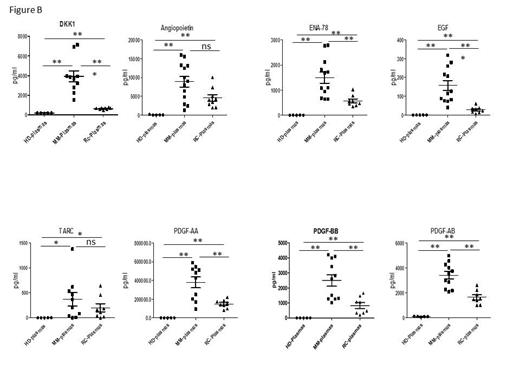
Legend to figure: Cytokine expression in MM, CR and HD-plasmas
(A) Representative images of cytokine array blots probed with the plasma samples. The red boxes identify the cytokines significantly dysregulated in MM- as compared to HD- or CR-plasmas and further analyzed by ELISA. The blue boxes identified the cytokines similarly expressed in MM-/CR-/HD-plasmas. (B) Cytokine concentrations in the HD-plasmas (n=5), MM-plasmas (n=11) and CR-plasmas (n=8) were measured by ELISA. * p < 0.05, ** p < 0.01, *** p < 0.001, ns (not significant).
Delhommeau: Novartis: Consultancy; BMS: Consultancy; Celgene: Consultancy. Garderet: Celgene: Consultancy; Janssen: Consultancy; Amgen: Consultancy; Sanofi: Consultancy; Takeda: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal